Veterans Affairs: Information on Protected Research Time for Clinician-Scientists
Fast Facts
At the Department of Veterans Affairs, clinician-scientists provide care to veterans and conduct research. These research efforts have led to important medical advancements, such as the first liver transplant. But high demand for patient care can make it hard to also do research.
In this Q&A we looked at VA's research program and found:
Protected research time helps recruit and retain top clinician-scientists
VA centers determine the amount of research time based on the research, funding, and patient needs
Rural VA centers have more difficulty protecting research time as they are likely far from academic centers that may support VA research

Highlights
What GAO Found
According to VA, the majority of the Department of Veterans Affairs’ (VA) nearly 4,000 VA researchers are clinician-scientists—those who conduct research and provide care to veteran patients. The dual role of clinician-scientists in VA’s health system allows them to identify the needs of veteran patients and then make discoveries that can move from the research setting to the patient care setting.
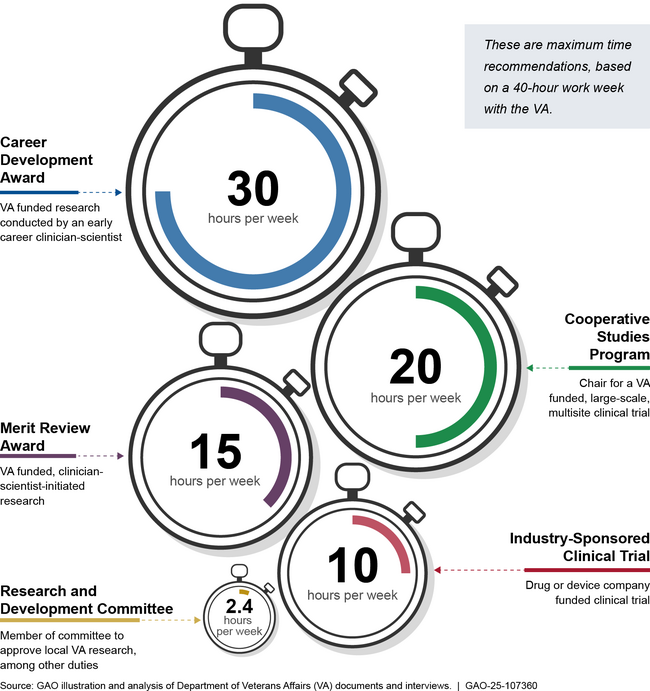
Providing clinician-scientists with time that they can dedicate solely to research (protected research time) helps VA recruit and retain top clinician-scientists. In particular, officials and clinician-scientists said VA’s Career Development Award, which provides up to 30 hours of protected research time per week to early career researchers, attracts young clinician-scientists and helps establish their research careers. Officials and clinician-scientists also said protected research time improves clinician-scientists’ productivity by increasing their ability to initiate research and by providing dedicated blocks of time in which they can focus on research.
VA medical center leaders determine the amount of protected research time to provide clinician-scientists by considering recommendations from VA’s Office of Research and Development (ORD) and other factors such as such as the type of research, project funding source, and patient care needs.
Examples of VA Guidance for Protected Research Time Provided to Clinician-Scientists
Why GAO Did This Study
For nearly 100 years, the VA’s research program has focused on enhancing the well-being of veterans and the nation through scientific discovery. However, leaders of VA medical centers and their clinical departments face difficult resource decisions in finding a balance between supporting research and providing patient care. The high demand for patient care may present a challenge to providing protected research time for clinician-scientists, particularly if they are working in a VA medical center that has staffing constraints.
The Consolidated Appropriations Act, 2023, includes a provision for GAO to review the amount of time dedicated for research by VA clinician-scientists. This report provides information on VA’s policies and practices for research time and how research time relates to clinician-scientist recruitment, retention, resource use, and productivity at selected VA medical centers.
GAO reviewed VA data on selected VA medical centers’ research funding, analyzed VA guidance and other documents, and interviewed officials from VA’s Office of Research and Development, Office of the Chief Operating Officer, Veterans Integrated Service Networks, and a non-generalizable sample of seven VA medical centers. GAO selected medical centers that provided diversity in research program size, geographic location, and program features such as areas of specialty. From these medical centers, GAO interviewed 36 clinician-scientists and 23 clinical chiefs from a variety of disciplines. GAO also interviewed each medical center’s leaders and officials from research offices. GAO collected data from each selected medical center on the amount of protected research time provided to clinician-scientists, but data were not available on the actual amount of time clinician-scientists spend on research or how they use their research time.
For more information, contact Candice N. Wright at (202) 512-6888 or WrightC@gao.gov.
